Over the years, the pharmaceutical industries have witnessed a remarkable transformation with the integration of Artificial Intelligence (AI) in medicine development and efficacy testing. The use of AI has revolutionized the way skincare medicines are formulated, dosed, and tested for stability and efficacy. AmWiner Brain Wise Research Institute became the first pharmaceutical manufacturing company to utilize the power of artificial intelligence tools in drug product development.
THE PROBLEM:
The Challenges in pharmaceutical drug formulation have increased significantly in recent decades, while at the same time the market situation has grown increasingly challenging. New chemical entities in the pipeline are facing solubility challenges, with about 80 percent of them reported as having poor solubility. Since formulation solubility is a critical prerequisite for good bioavailability and therapeutic efficacy, finding ways to improve solubility without changing the chemical nature of the NCE has never been more important.
In addition, the current industry trends towards regionalization and personalization of treatment approaches necessitate faster, more efficient, and increasingly flexible solutions. As a result, continuous pharmaceutical manufacturing is currently in a prominent field of development, driving the need for tighter process controls and more thorough process and product understanding. An intelligence technology having access to a wide range of suitable excipients is a prerequisite for a successful formulation approach, and this is what we offer to all pharmaceutical industries.
.Drug formulation development challenges for small biopharmaceutical companies are becoming increasingly important as drivers of innovation in drug development. It has recently been estimated that most drugs currently in development are in the hands of small biopharmaceutical companies. Formulation is one of the most crucial parts of the drug development process. We must be able to deliver the drug to the body in whatever dosage form necessary that provides the best effect for that medication. Without proper formulation that guarantees solubility and bioavailability, patients’ health and healthcare system will be at tremendous risk. The small pharmaceutical drug companies want to quickly develop formulations for clinical trials, at the same time, but they have reduced resources and technology challenges to develop the most cost- effective formulation as quickly as possible, the quicker the medication can make its way to patients, the faster we can impact the worlds healthcare system.
.The two parameters; time and cost are the biggest challenges in formulation development, especially of small molecules. This means how well that formulation releases the molecule and how well that molecule passes from the gastrointestinal tract into the systemic circulation will determine the therapeutic outcome. And this process is less efficient when drug solubility is reduced. And what we’ve seen over the past decades is that the solubility of drug molecules has been decreasing on yearly basis, that up to 80% of the drugs in the drug development pipeline are poorly soluble. Poor soluble drugs in the system means longer patient recovery time. An intelligence technology having access to a wide range of suitable excipients is a prerequisite for a successful pharmaceutical drug developer.
.Small pharmaceutical companies have used a variety of approaches to address these drug formulation challenges, including the use of new technical platforms, the use of new formulations or technologies that enhance the actions of known drugs. One example is a new, thermostable polymer for hot-melt extrusion processes that combines stabilization of the solid amorphous form in the tablet and inhibition of drug precipitation in dissolution. Another formulation technology being used is a silica-based drug carrier that features a functionalized particle structure that allows for API adsorption onto its surface and pores, stabilizing the drug in a soluble amorphous form. These two methods have not yielded a definitive success that the need for intelligent technology capable of guaranteeing effective formulation will help strengthen and improve our healthcare system and patients’ wellbeing.
THE SOLUTION WE OFFER IS: AI-PHARMA
.Over the years, AmWiner & Raphe Holdings forecasted a remarkable transformation of pharmaceutical industry in drug development and patient care with the integration of Artificial Intelligence (AI) in medicine development and efficacy testing. The use of AI will revolutionize the way drug products are formulated, dosed, and tested for stability and efficacy.
.Understanding AI PHARMA Technology is a field of artificial intelligence (AI) in which software learns from imputed data to perform a task throughout the drug development and manufacturing process that will increase drug efficiency and effectiveness, decreasing the time and cost required to bring new drugs to market. Giving small pharmaceutical companies the opportunity to be part of the manufacturing process of bringing new drugs with optimum solubility and bioavailability to the market and increasing the supply of the existing ones. These improvements will save lives and reduce suffering by getting drugs to patients in need more quickly and could allow researchers to invest more resources in areas such as rare or orphan diseases.
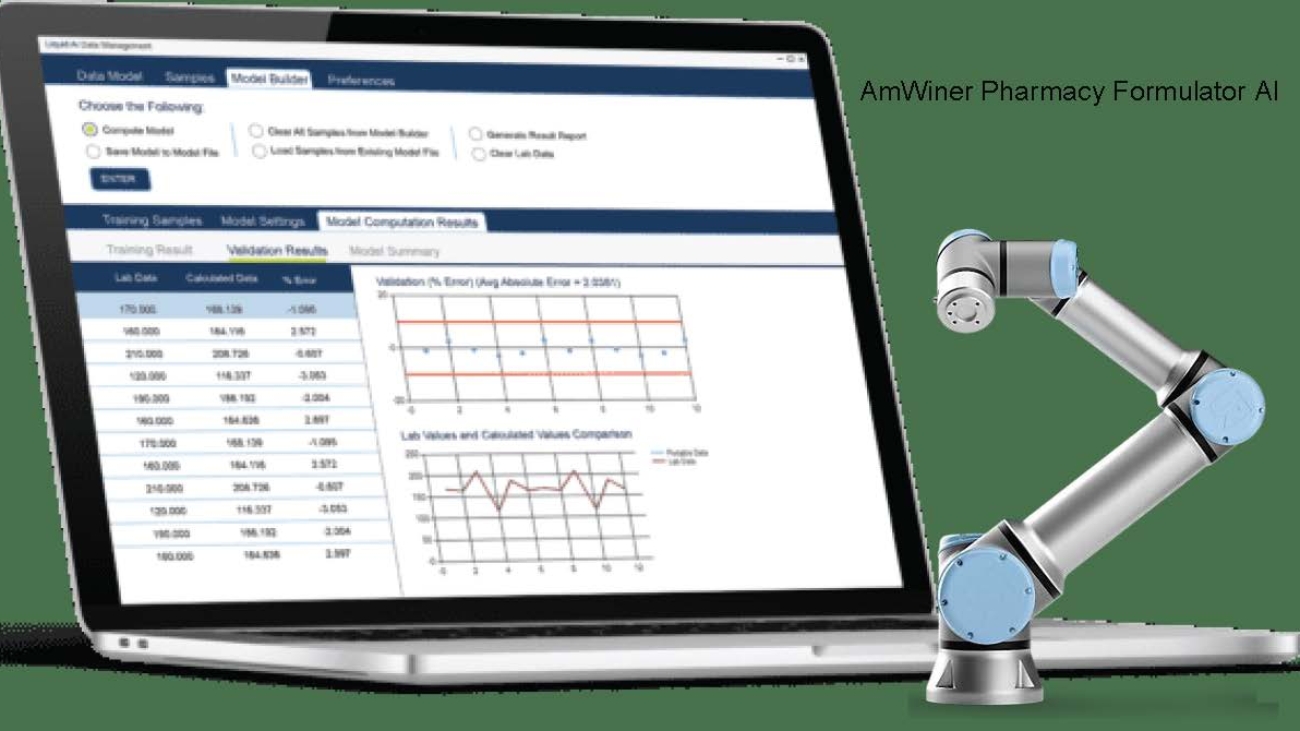
AmWiner`s .AI PHARMA Technology significantly enhance the process of medicine development by taking the guesswork out of every formulation. The chemist/pharmacist will know with 100% certainty the best and accurate percentage of Active Pharmacological Ingredient/s and expedient percentages to use his/her formulation that will yield the desired expected results, taking the human error of miscalculation that contributes to some adverse effects. When confronted with a custom formula, you`re confident that the technology will determine the accurate percentage of the active and expedients needed to use based on the imputed objective you intend to achieve. Even when you add any ingredient more or less of what is required, the “AI PHARMA Technology” indicates which ingredient/s you have in excess and what to do to balance your formulation, expediting the formulation and optimizing the dosages.
.Through advanced algorithms of the AI PHARMA technology for pharmaceutical industries analyzes the finished product to produce a more accurate certificate of analysis and SDS document, conduct stability product testing, shelf-life test, and efficacy testing. AmWiner’s “AI PHARMA Technology” takes the guesswork out when conducting Quality control testing on solid oral dosage tablets/capsules, friability tests, tablet disintegration tests, dissolution tests, and moisture content tests. This tool is a power engineer in tablet coating when manufacturing XR or enteric-coated pills. It determines the exact percentage of the coating ingredient required and should show the best coating substance suitable for each tablet moiety. You (the chemist) can take the drying and chipping issues of the coating process out of your list of worries because this technology delivers 100% accurate coating technique and guide for your OSD batches.
.AmWiner’s “AI PHARMA Technology” collects vast amounts of data related to human biology, dermatological conditions, and ingredient interactions to identify potential compounds, ingredients, and therapeutic interventions necessary for every skincare or bodily concern. This not only accelerates the research and development phase but also enables the creation of highly effective and personalized solutions. Moreover, these AI algorithms predicts the performance of different formulations, leading to the creation of products that offer superior outcomes in addressing various patients’ skin concerns.
.The integration of AmWiner AI PHARMA Technology” in pharmaceutical product development facilitates the identification of novel compounds with therapeutic potential. By analyzing molecular structures and biological pathways, these AI algorithms pinpoint promising treatments for any skincare or bodily issues, thus streamlining the therapeutic process. This approach opens new avenues for developing innovative treatments for common skin and body ailments, such as acne, vitiligo, rosacea, pigmentation, eczema, and aging-related conditions and enable researchers to assess the safety and efficacy of potential drug products, fostering the creation of products that are not only effective but also safe for prolonged use.
.In essence, AmWiner AI PHARMA Technology” revolutionizes pharmaceutical drug development by empowering researchers to navigate the complexities of human biology and formulation optimization with unprecedented speed and precision. The insights generated by the algorithm will pave the way for the creation of next- generation drug products that cater to diverse health conditions, marking a paradigm shift in the field of dermatology and general medicine.
The Role of AmWiner AI PHARMA Technology” in Medicine Dosing. In the realm of drug product development, accurate dosing is paramount to ensure optimal therapeutic outcomes and minimize adverse effects. AmWiner Pharmacy AI technology” plays a pivotal role in precision dosing by leveraging data analytics and predictive modeling to determine the most effective dosage regimens for any drug product. By analyzing patient-specific factors, such as skin type, age, and medical history, the AmWiner AI algorithms can tailor dosing recommendations to individual needs, thereby enhancing the efficacy and safety of treatments. This technology adapts to real-time changes in patient responses and adjust dosage parameters accordingly. This dynamic dosing approach enables the healthcare professionals to fine-tune treatment plans based on evolving patient conditions, ultimately optimizing the therapeutic benefits of the medicines. Additionally, the AI algorithms identify potential drug interactions and contraindications, offering valuable insights to healthcare providers and patients alike.
.By harnessing the power of AmWiner AI PHARMA technology” in medicine dosing, the pharmaceutical industry is poised to deliver personalized treatment regimens that are tailored to everyone’s unique physiological and dermatological profile. This not only maximizes the efficacy of the drug product but also minimizes the risk of adverse reactions, setting a new standard for precision and safety in dermatological care.
.Utilizing AmWiner “AI PHARMA Technology” for Product Stability Testing. Ensuring the stability of any drug product is essential to maintain its efficacy and safety over time. This AI technology emerge as a game-changer in product stability testing, enabling manufacturers to assess the impact of various environmental factors, storage conditions, and formulation changes on the shelf-life and performance of skincare products. Through predictive modeling and accelerated testing methodologies, the algorithms predict the degradation kinetics of active ingredients, anticipate potential formulation instabilities, and optimize packaging materials to prolong product shelf-life.
.The AI-powered stability testing moiety allows for the rapid identification of formulation modifications that can enhance product stability without compromising efficacy. By simulating different storage scenarios and analyzing real-time data from stability studies, these AI algorithms recommend adjustments to formulation parameters, packaging designs, and storage protocols to mitigate degradation risks and extend the lifespan of drug products. This proactive approach to stability testing minimizes product wastage and ensures consistent efficacy throughout the product’s shelf-life, thereby enhancing customer satisfaction and brand reputation.
. Furthermore, it facilitates the early detection of stability issues by flagging potential risks based on predictive analytics and historical data. This proactive monitoring capability empowers drug product manufacturers to preemptively address stability concerns, implement corrective measures, and maintain the integrity of their products throughout their lifecycle. As a result, the AI-driven stability testing not only streamlines the product development process but also fosters the creation of high- quality drug products that meet stringent stability and performance standards.
.Incorporating AmWiner AI PHARMA Technology” in product development: Best practices. To maximize the benefits of this AI, compounding pharmacist and chemists will prioritize data quality and diversity, ensuring that the AI algorithms collect comprehensive datasets that encompass a wide range of demographics, and dermatological conditions. This inclusive approach to data collection and analysis will foster the creation of effective drug products across diverse populations and demographics, promoting inclusivity and accessibility in healthcare systems. By fostering partnerships between dermatologists, pharmacologists, data scientists, and AI experts, drug product developers can leverage diverse expertise to optimize formulation strategies, dosing protocols, and efficacy testing methodologies. This collaborative approach not only enriches the insights generated by the AI algorithm but also ensures that drug products are developed with a comprehensive understanding of human biology, therapeutic mechanisms, and patient needs.
. Continuous validation and refinement of Amwiner`s AI PHARMA technology models are critical to maintaining the accuracy and reliability of drug development processes. The chemist/pharmacist will regularly validate the AI algorithms against real-world clinical data, patient outcomes, and market feedback to ensure that the insights and recommendations offered by AmWiner remain aligned with actual patient needs and treatment outcomes.
.To develop a drug product with high precision and efficacy or to explore cutting-edge solutions in dermatology, call us at: 1-469-428-5505 or email [email protected] visit our website www.Healthcareprivatelabel.com or www.Rapheceutique.com